Pregnancy is usually a journey full of anticipation, but for some women, obstetric antiphospholipid syndrome (OAPS) strikes like an unexpected storm—recurrent miscarriage, fetal growth restriction, severe preeclampsia, and other adverse outcomes leave 20%–30% of patients facing difficulties even with standard treatment.
Recently, a landmark study published in Advanced Healthcare Materials brings new hope to patients with refractory OAPS! A research team from Shandong University used multi-dimensional verification including animal models, placenta chips, and molecular experiments to confirm for the first time that simvastatin can reverse placental hypoperfusion and trophoblast dysfunction caused by OAPS by activating the KLF2/eNOS signaling pathway, making it a promising novel therapeutic agent for this condition.
Ⅰ.Why Is OAPS So Dangerous?
OAPS is an autoimmune disease driven by antiphospholipid antibodies (aPLs). Its core harms are:
- Antibodies attack placental trophoblast cells, leading to impaired spiral artery remodeling, vascular stenosis, and inflammatory infiltration;
- Abnormal uteroplacental hemodynamics characterized by high resistance and low perfusion, resulting in insufficient oxygen and nutrient supply to the fetus;
- Reduced invasive capacity of trophoblast cells, failing to form a healthy maternal-fetal circulatory interface, and ultimately leading to adverse pregnancy outcomes.
Conventional treatment relies on low-dose aspirin plus low-molecular-weight heparin, yet a large number of patients do not respond to this therapy, creating an urgent need for more effective treatment strategies and clear mechanistic support.
Ⅱ.New Scientific Breakthrough: The Life-Saving Mechanism of Simvastatin
The research team innovatively combined an OAPS mouse model with placenta chip technology to dissect the therapeutic effects of simvastatin step by step:
1. Improves Pregnancy Outcomes and Repairs Vascular Damage
In the OAPS mouse model, simvastatin significantly reduced fetal resorption rate and increased fetal weight, while alleviating pathological injuries such as decidual vascular hyperplasia, wall thickening, and fibrinoid necrosis. Immunoassays showed it effectively inhibited the overexpression of pro-inflammatory cytokines (IL-1β, IL-6) and adhesion molecules (VCAM-1, ICAM-1), reducing vascular endothelial inflammation.
2. Corrects Hemodynamics and Enhances Placental Perfusion
Ultrasound examination confirmed that simvastatin reduced the pulsatility index (PI) and resistance index (RI) of the uterine artery in OAPS mice, lowering vascular resistance. It also restored endothelial nitric oxide synthase (eNOS) expression, improving vasodilation and ensuring adequate blood supply to the placenta.
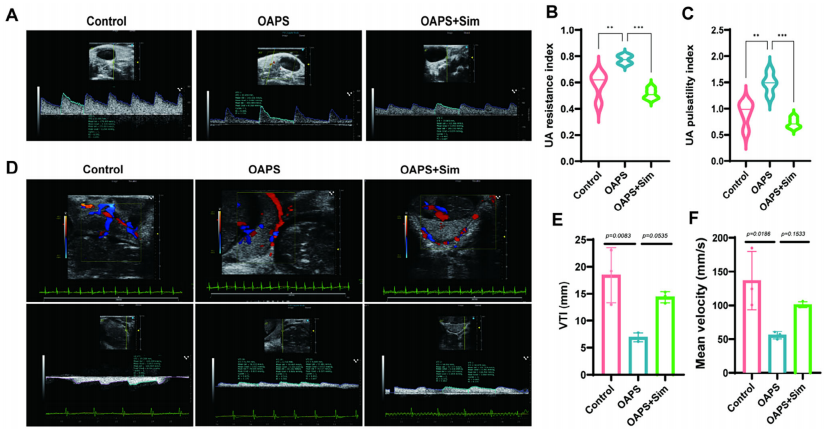
Simvastatin improves uterine artery hemodynamics in mice with obstetric antiphospholipid syndrome. (A) Ultrasound localization of the placental uterine artery and representative color Doppler and pulsed-wave Doppler spectrograms in each group; (B, C) Quantitative analysis of uterine artery blood flow parameters—pulsatility index (PI) and resistance index (RI) (n = 4, one-way ANOVA); (D) Color Doppler and pulsed-wave images of blood flow at the uterine artery branch level; (E, F) Statistical analysis of blood flow parameters—velocity time integral (VTI) and mean flow velocity of uterine artery branches (n = 3, one-way ANOVA)
3. Placenta Chip Validation: Reverses Trophoblast Damage Under High Shear Stress
To simulate the early pregnancy maternal-fetal interface microenvironment, the team built a placenta chip containing extravillous trophoblasts (EVT) and human umbilical vein endothelial cells (HUVEC). The results revealed:
- High shear stress significantly suppresses EVT invasion—a key pathological feature of OAPS;
- Simvastatin promotes EVT invasion indirectly by upregulating eNOS, suppressing inflammation, and restoring matrix metalloproteinase 2 (MMP2) levels, creating conditions for spiral artery remodeling.
4. Core Mechanism: Activation of the KLF2/eNOS Signaling Pathway
Transcriptome sequencing and molecular experiments identified the critical pathway: antiphospholipid antibodies (aPLs) inhibit KLF2 (Kruppel-like factor 2) and eNOS expression in endothelial cells, and simvastatin reverses this suppression. Through the KLF2/eNOS axis, it maintains endothelial cell homeostasis and improves placental function at the root.
Ⅲ. Key Tool for Successful Experiments: High-Quality ELISA Kits
Accurate experimental results rely on reliable detection tools. In this study, quantitative detection of inflammatory factors (IL-6, IL-1β) and matrix metalloproteinase 2 (MMP2) was performed using Reed Biotech ELISA kits. Their stable performance provided critical data for mechanistic validation:
- Human High-Sensitivity IL-6 ELISA Kit (Cat. No.: RE3186HG)
- Human IL-1β ELISA Kit (Cat. No.: RE1074H)
- Human MMP-2 ELISA Kit (Cat. No.: RE1838H)
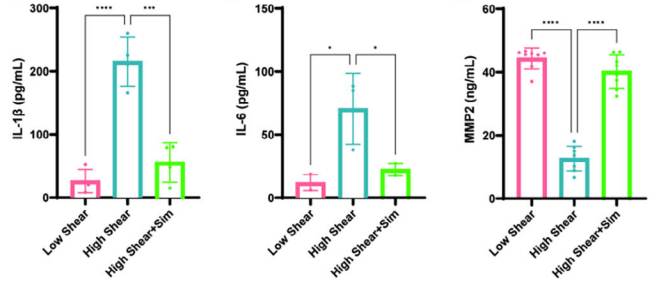
Enzyme-linked immunosorbent assay (ELISA) for the concentrations of interleukin-1β, interleukin-6, and matrix metalloproteinase 2 (MMP-2) in the outflow fluid of the detection channel (n = 3, one-way ANOVA)
High-quality ELISA kits are indispensable for researchers, supporting precise analysis of pathological processes in both basic mechanistic research and clinical biomarker detection.
Ⅳ .Clinical Significance and Future Outlook
This study not only reveals the core mechanism of simvastatin in treating OAPS but also establishes an in vitro model close to the physiological state via placenta chip technology, providing a new paradigm for drug screening in pregnancy-related diseases. Leveraging its anti-inflammatory and hemodynamic-improving properties, simvastatin is expected to become a combination therapy option for refractory OAPS in the future.
