Interpretation of Latest Findings in Scientific Reports: How Extracellular Vesicles Record the Heart’s "Stress Memory"
Have you heard of Takotsubo Syndrome (TS), also known as "broken heart syndrome"? Triggered by extreme emotional stress or physical strain, this condition mimics acute myocardial infarction, characterized by temporary dysfunction of the cardiac apex (apical ballooning), yet coronary arteries are typically unobstructed.
Scientists have long puzzled over why different regions of the heart respond so differently to the same surge of stress hormones. A recent study published in Scientific Reports used proteomic techniques to uncover, for the first time, region-specific molecular changes in extracellular vesicles (EVs) in broken heart syndrome, opening a new window into understanding this mysterious disease.
Key Finding: Distinct "Communication Profiles" Across Cardiac Regions
A research team from the University of Gothenburg successfully induced broken heart syndrome in a high-fidelity rat model. Twenty-four hours after onset, they isolated and analyzed tissue-derived extracellular vesicles (EVs) from the affected apex and unaffected base of the heart.
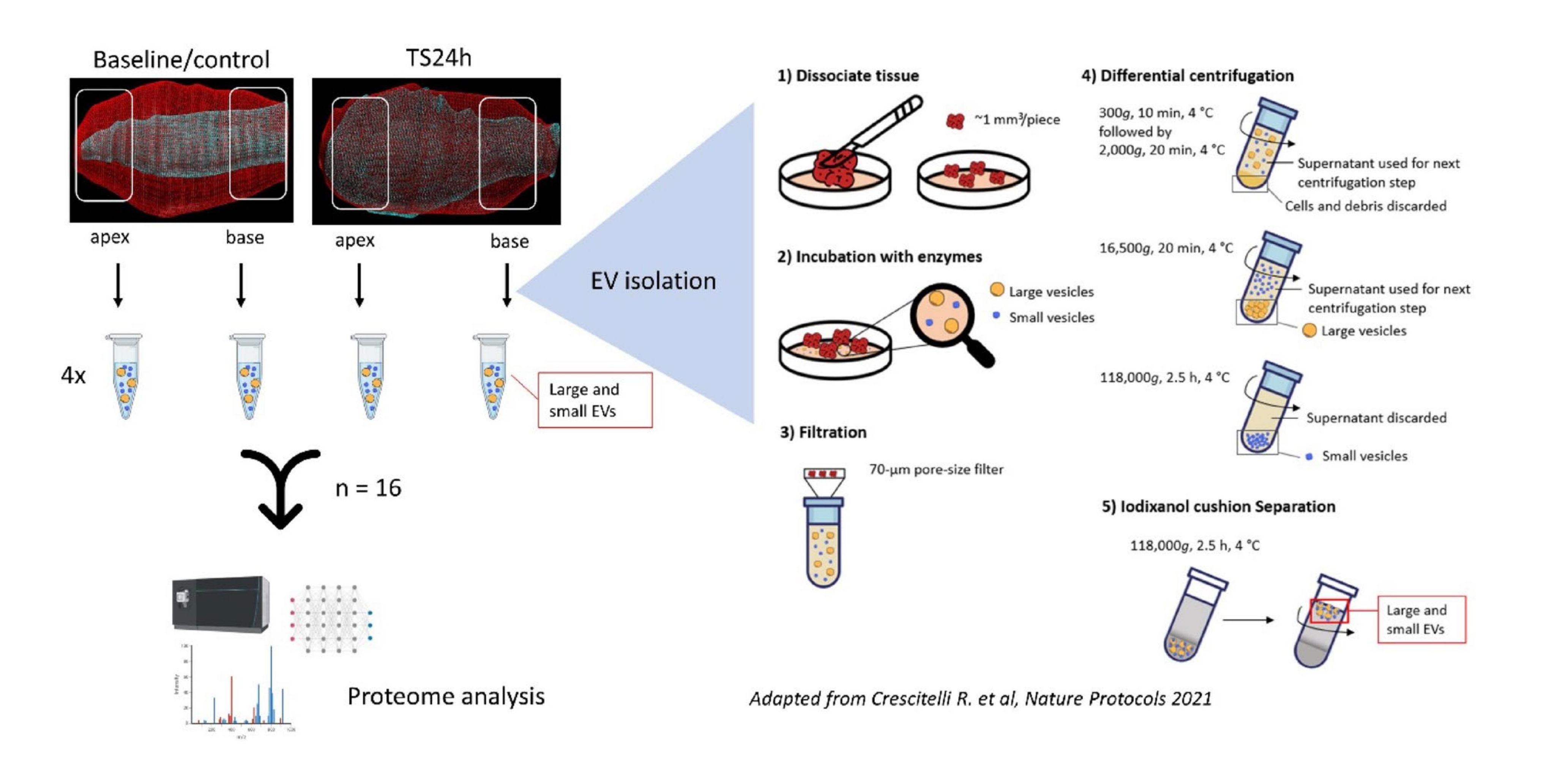
Key Data
- 2,093 proteins were successfully identified.
- 238 differentially expressed proteins were found in the apex compared with controls.
- A total of 562 differentially expressed proteins were detected between the diseased apex and healthy base.
These results confirm that the affected apical region has a unique molecular adaptive mechanism, with a distinctly different protein profile in its extracellular vesicles compared to other cardiac regions.
Molecular "Contrast" Between Regions
Functional enrichment analysis of the protein cargo in these extracellular vesicles revealed several critical biological process alterations:
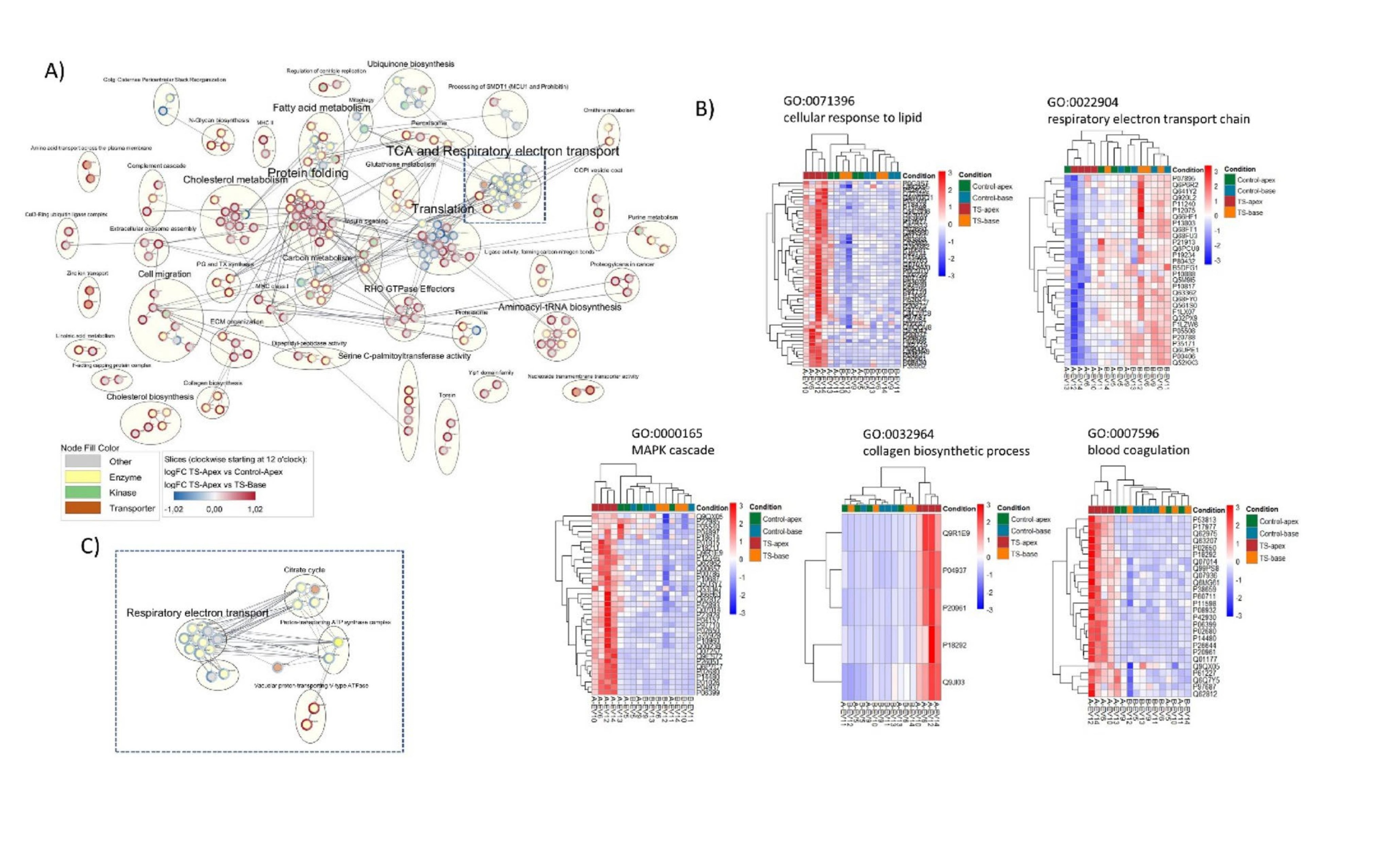
1. Enhanced Immune and Inflammatory Response
In the injured apex, proteins related to immune response and tissue repair were significantly upregulated. For instance, increased levels of complement system–activating proteins such as Ficolin-2 indicate active local inflammatory regulation—marking not only injury but also the heart’s initiation of self-repair signals.
2. Suppressed Mitochondrial Function
In sharp contrast, proteins linked to mitochondrial function and energy metabolism (e.g., key components of Complexes I and IV) were markedly decreased. This aligns with clinical observations of impaired myocardial energy metabolism (reduced PCr/ATP ratio) in the acute phase of broken heart syndrome, explaining why the myocardium temporarily "stops working."
3. Activation of Cell Survival Signaling Pathways
Enrichment of proteins associated with the MAPK signaling pathway was also observed. This likely represents a cytoprotective and survival mechanism activated by the heart under extreme stress, aiming to prevent cell death and support subsequent functional recovery.
Notably, unlike the increased extracellular vesicle release commonly seen in myocardial infarction and other cardiovascular diseases, this study found significantly fewer extracellular vesicles and lower total protein content in the injured apex of broken heart syndrome.
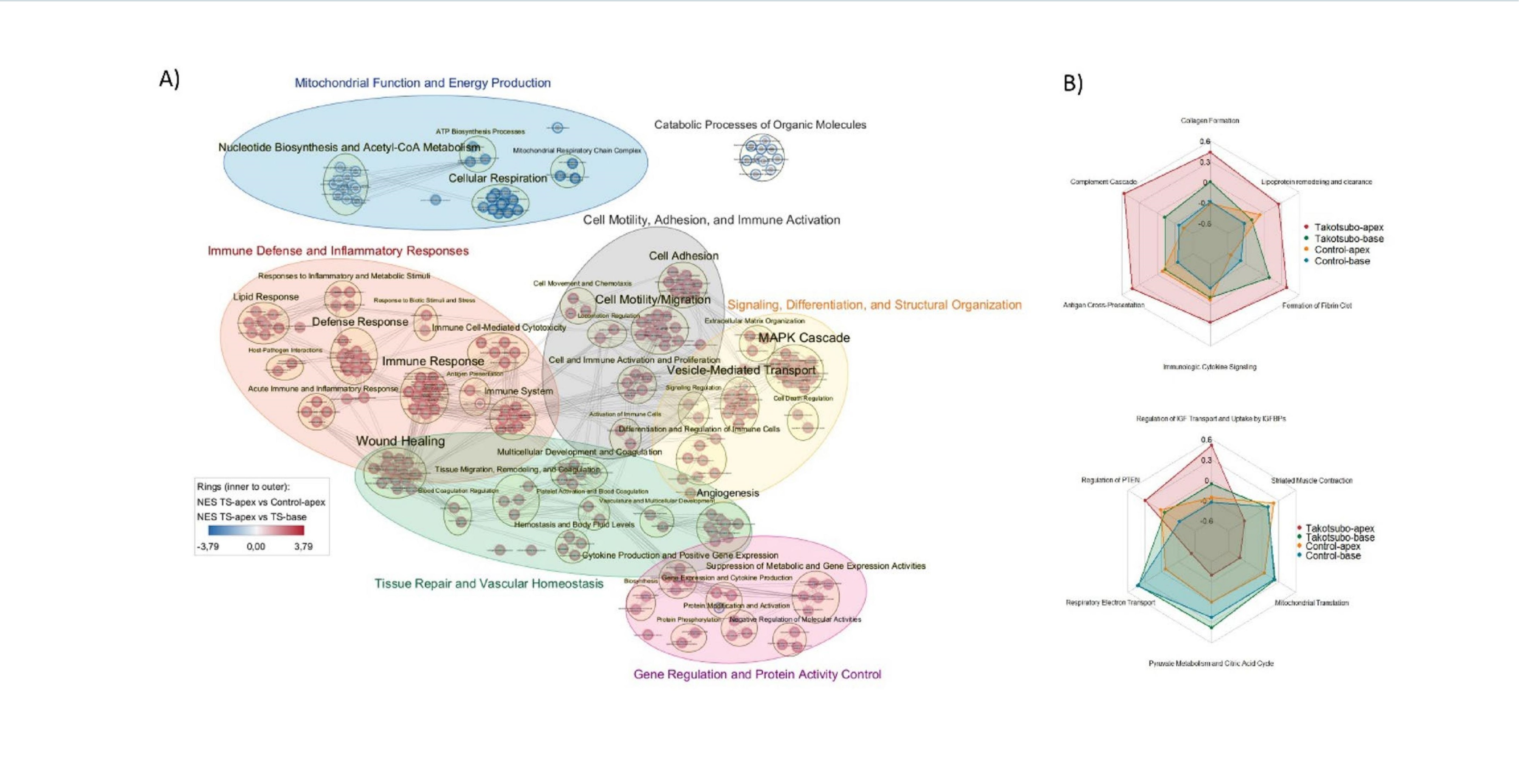
"This phenomenon may reflect a unique pathophysiological mechanism of broken heart syndrome. It could result from reduced local vesicle biogenesis, or structural changes under stress impairing vesicle recycling efficiency," the research team speculated.
Research Significance and Future Outlook
This study is the world’s first proteomic characterization of cardiac tissue–derived extracellular vesicles in broken heart syndrome. It not only confirms the high regional specificity of the condition but also provides potential diagnostic biomarkers and therapeutic targets.
Future research will focus on:
- Dynamically monitoring vesicle changes at different time points to map the full course of disease progression.
- Exploring how these vesicles mediate intercellular communication in the heart to drive repair or injury.
- Validating whether these specific proteins can serve as noninvasive clinical detection markers.
While "broken heart" sounds emotional, science is steadily unraveling its rational biological logic. Every molecular fluctuation is a signal of the heart striving to heal itself.
Reference
Ermir Zulfaj et al. "Proteomic analysis of tissue-derived extracellular vesicles shows region-specific molecular changes in a rat model of takotsubo syndrome." Scientific Reports (2026). DOI: 10.1038/s41598-026-42812-5
