Hypoxia is the most prevalent and lethal microenvironmental characteristic within solid tumors. The central executor of cellular responses to hypoxia is a class of key transcription factors known as HIF-1, which governs cancer cell survival, metabolism, angiogenesis, metastasis, and drug resistance, emerging as an undisputed focus in basic and translational cancer research in recent years. This article deciphers the structure, regulation, functions, and clinical value of HIF-1 in a professional yet accessible manner.
I. What is HIF-1? The Core Transcription Factor for Hypoxia Response
Hypoxia-inducible factor -1 (HIF-1) is a heterodimeric protein belonging to the bHLH-PAS family, consisting of an oxygen-sensitive α subunit and a constitutively expressed β subunit (HIF-1β/ARNT). First identified in 1992 as a key protein regulating erythropoietin (EPO), HIF-1 was later found to act as the master switch for cells to sense low oxygen levels and initiate adaptive programs.
II. Structure and Isoforms: Three Isoforms with Distinct Functions
The HIF family comprises three isoforms,HIF-1, HIF-2 and HIF-3, among which HIF-1α and HIF-2α play dominant roles in tumor progression. Their shared structural domains are critical to their functions, including:
• bHLH: Mediates binding to hypoxia response elements (HRE) on DNA
• PAS: The core binding site for heterodimerization between the α and β subunits
• ODDD: Oxygen-dependent degradation domain that determines protein degradation
• N-TAD/C-TAD: Binds coactivators such as p300/CBP to initiate transcription
• NLS: Nuclear localization signal that mediates the translocation of the complex into the nucleus
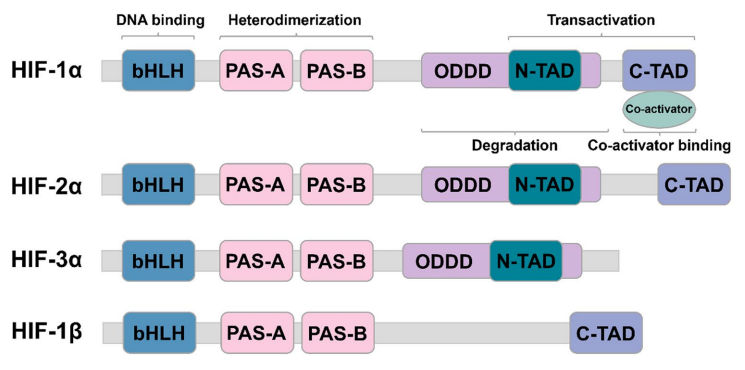
HIF subtype structure and domain [from Shi,et.al].
|
Isoform |
Expression Characteristics |
Core Functions |
|
HIF-1α |
Ubiquitous expression throughout the body |
Acute hypoxia response; master regulation of glycolysis and cell survival |
|
HIF-2α |
Tissue-specific expression in the kidney, lung, blood vessels, etc. |
Chronic hypoxia adaptation, vascular remodeling, stemness maintenance, and cell proliferation |
|
HIF-3α |
Restricted distribution |
Negative regulation; inhibition of HIF-1/2 activity |
III. Dimerization and Nuclear Translocation: How Do α and β Subunits Assemble and Enter the Nucleus?
HIF-1 activation proceeds through three essential steps: dimerization → nuclear translocation → transcription, which represents the core molecular logic of this pathway.
Following stabilization of HIF-1α in the cytoplasm, the PAS domain of the α subunit binds to that of the β subunit to form a stable heterodimer. After assembly in the cytoplasm, the α/β heterodimer is recognized by nuclear transport proteins via the nuclear localization signal (NLS) on the α subunit, and is actively translocated into the nucleus through the nuclear pore complex. Upon nuclear entry, it binds to HRE sequences and initiates the transcription of hundreds of downstream genes. In simple terms: the α subunit senses hypoxia and leads the complex into the nucleus, while the β subunit stabilizes the structure and supports transcription – both are indispensable for successful transcriptional initiation.
IV. Canonical Regulation: Oxygen Dictates the Fate of HIF-1α
The core regulatory logic of HIF-1α is straightforward: it is degraded under normoxic conditions and activated under hypoxic conditions.
Under normoxia (sufficient oxygen), HIF-α is hydroxylated by PHD enzymes, ubiquitinated and labeled by VHL, and rapidly degraded by the proteasome; FIH further blocks coactivator binding, resulting in complete silencing of the pathway. In a hypoxic environment, the activity of PHD/FIH is inhibited, preventing HIF-1α degradation and leading to its cytoplasmic accumulation. The PAS domain of accumulated HIF-α then binds to the β subunit to form a heterodimer, which attaches to hypoxia response elements (HRE) and recruits coactivators such as p300/CBP to the promoter regions of target genes, thereby regulating gene transcription. This is the fundamental reason for the persistent activation of HIF in hypoxic tumors.
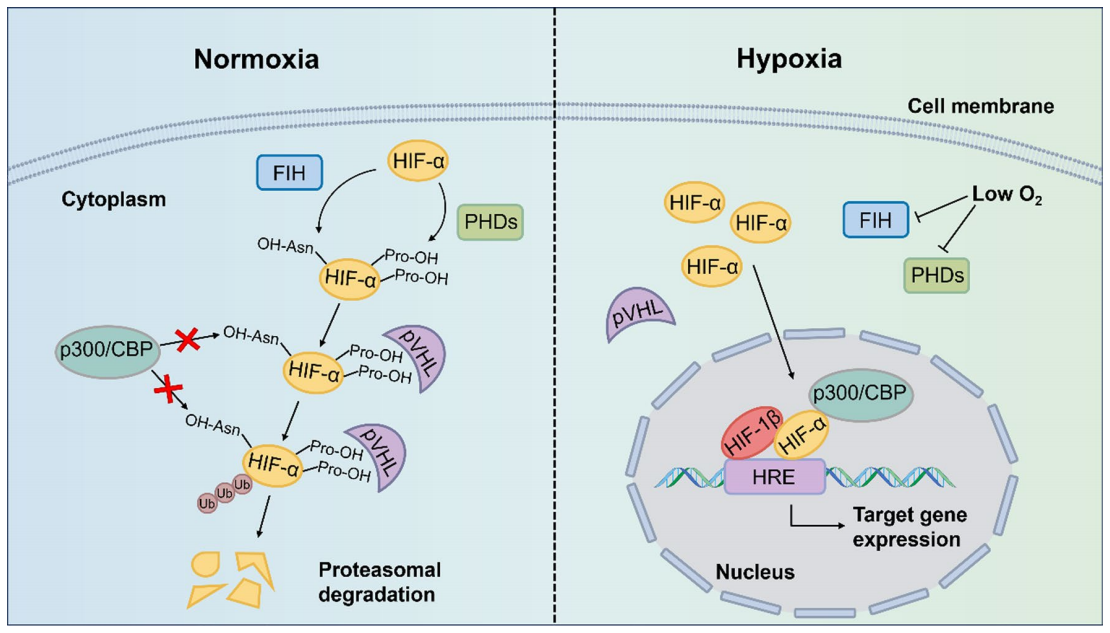
Classical regulation of HIF-α(HIF-1α/HIF-2α) activity under normoxic and hypoxic conditions [from Shi,et al].
V. Non-Canonical Regulation: HIF Activation Independent of Hypoxia
HIF activation is not entirely dependent on hypoxic conditions, but can also be achieved through multiple non-canonical regulatory pathways. Recent studies have revealed that loss-of-function mutations in tumor suppressor genes (VHL, p53, and PTEN) directly lead to the stable accumulation of HIF protein; aberrant activation of oncogenic signaling pathways such as PI3K/AKT and MAPK/ERK also significantly promotes HIF expression and activity. Meanwhile, post-translational modifications including phosphorylation, acetylation, SUMOylation, and the newly discovered lactylation (at mouse K644 and human K12 residues) can enhance HIF-1α stability by blocking VHL binding. In addition, inflammatory cytokines such as IL-1β and TNF-α can upregulate HIF expression. These non-canonical mechanisms collectively explain the key reason for high HIF expression in some tumors even under non-hypoxic conditions.
VI. The Oncogenic Roles of HIF in Tumors
HIF exerts a core regulatory role in tumor progression and extensively participates in the malignant biological behaviors of tumors by activating downstream target genes: HIF-1α primarily mediates metabolic reprogramming in tumor cells, upregulating key glycolytic enzymes and glucose transporters to maintain energy supply for cancer cells in the hypoxic microenvironment; it also promotes the expression of angiogenesis-related genes such as VEGF and Ang, driving the formation of tumor neovascularization. Furthermore, HIF regulates cell motility, extracellular matrix remodeling, and epithelial-mesenchymal transition (EMT) programs, enhancing the invasive and metastatic potential of tumor cells. It mediates resistance to radiotherapy and chemotherapy by reducing DNA damage, upregulating drug efflux pumps, and maintaining the characteristics of cancer stem cells; it also inhibits immune cell infiltration and cytotoxicity by remodeling the tumor immune microenvironment. Ultimately, these effects collectively promote malignant tumor progression and treatment resistance.
VII. Clinical Significance: From Biomarker to Targeted Therapy
Clinically, HIF-1α/2α not only serve as important prognostic biomarkers for tumors – their high expression usually indicates greater tumor invasiveness, increased metastatic potential, and poorer patient prognosis – but also represent highly promising therapeutic targets. Belzutifan (MK-6482), the world's first HIF-2α-specific inhibitor, has been approved for the treatment of VHL syndrome-associated renal cell carcinoma, and a number of broad-spectrum HIF inhibitors are in early-stage clinical research. Additionally, the newly discovered lactylation modification, as a cross-regulatory node between metabolic and hypoxic pathways, provides a completely new direction and strategy for combined tumor therapy.
References:
1. Shi Y, Gilkes DM. HIF-1 and HIF-2 in cancer: structure, regulation, and therapeutic prospects. Cell Mol Life Sci. 2025;82(1):44. Published 2025 Jan 18. doi:10.1007/s00018-024-05537-0
2. Lima JDCC, Hooker M, Li R, et al. HIFα isoform specific activities drive cell-type specificity of VHL-associated oncogenesis. Nat Commun. 2025;16(1):9185. Published 2025 Oct 16. doi:10.1038/s41467-025-64214-3
3. Li, C., Fu, C., Zhou, W. et al. Lactylation modification of HIF-1α enhances its stability by blocking VHL recognition. Cell Commun Signal 23, 364 (2025). doi:10.1186/s12964-025-02366-x
4. Zhang M, Li L, Mao X, et al. HIF-1α Drives Cellular Senescence Via Autophagy Activation to Promote AKI-to-CKD Transition. Am J Nephrol. Published online March 16, 2026. doi:10.1159/000551542
